Contact
Positions
Professor Emeritus
- Organization:
- West Virginia University School of Medicine
- Department:
- Pathology, Anatomy and Laboratory Medicine
- Classification:
- Faculty
Education
- PhD, Michigan State University
Publications
Selected Publications:
- A.K. Salm, B.E. Lally, E. Borysiewicza, D. Fil and G. Konat (2015) Analysis of extinction acquisition to attenuated tones in prenatally stressed and non-stressed offspring following auditory fear conditioning. Physiol. and Behav.. 139 (16): 157-166.
- A.K. Salm and J.L. Culberson (2015) Peer presentations of case studies. Medical Science Educator. 25 (4): 407–412.
- Bruner NR, Salm AK, Anderson KG. Effects of prenatal stress on lever-press acquisition with delayed reinforcement in male and female rats Behav Processes (2012 Mar) 89(3): 256-63.
- Konat GW, Lally BE, Toth AA, Salm AK. Peripheral immune challenge with viral mimic during early postnatal period robustly enhances anxiety-like behavior in young adult rats. Metab Brain Dis (2011 Sep) 26(3): 237-40.
- Kaur G, Salm AK. Blunted amygdalar anti-inflammatory cytokine effector response to postnatal stress in prenatally stressed rats. Brain Research (2008), 1196:1-12.
Link to PubMed for full list of publications: https://www.ncbi.nlm.nih.gov/pubmed/?term=AK+Salm
Research Interests
Adrienne K. Salm: Emerita Activities
Today my interactions with, and contributions to, the research enterprise are largely through invited editorial roles with various journals. When fully funded (thank you NSF and NIH) my laboratory, active for 30 years, pursued two major lines of research that largely underlie my areas of expertise. This, of course, is in addition to teaching 30 years of Medical Neurobiology lectures and laboratories, as well as other clinically oriented courses.
My foremost area of research was in the contribution of astrocytes to brain function and plasticity. The physical presence of astrocytes in the CNS serves to regulate communication among specific sets of neurons by modulating synaptic contacts, and through the establishment and maintenance of neuronal subgroupings. Astrocytes do this by undergoing structural plasticity, i.e., actively rearranging their processes to selectively segregate neuronal elements. They express a pantheon of receptors to CNS neurotransmitters to do so. We extended our study of glial cells to include microglia. While the early years were devoted to phenomena surrounding events in the hypothalamus we eventually also evaluated the role of astrocytes in events surrounding prenatal stress in the amygdala.
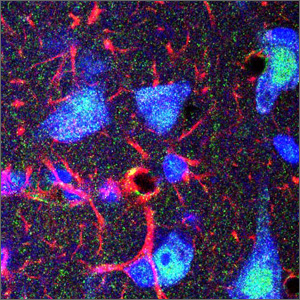
Figure 1. Astrocytes and neurons in the amygdala as seen by immunofluorescence for the glial fibrillary acidic protein (astrocytes:red) and Nissl substances (blue).
A second line of research was endeavoring to understand how the brain’s limbic system, particularly the amygdala, plays a role in how humans develop mental health conditions related to fear, i.e., anxiety disorders. These include conditions exacerbated by stress such as posttraumatic stress disorder, generalized anxiety disorder and social anxiety disorder. We used an animal model to examine behavioral, biochemical and structural changes in the brains of adult rats whose mothers were subjected to mild stress during pregnancy. Like humans with histories of childhood trauma, these animals were behaviorally hyperresponsive to stress and had altered brain biochemistry. We were able to show they also have an enlargement in the lateral subnucleus of the amygdala, which is the brain’s input area for sensory information concerning the external environment.
In the two years prior to my retirement, I had the good fortune to work on a new and exciting line of epidemiological research which examined the incidence of vascular dementia in West Virginia counties where Mountaintop Mining (MTM) had or had not occurred over the previous 15 years. We found a highly significant, dramatic, increase in both rates and incidence of dementia in the MTM counties. Our results were congruent with a growing awareness of the impact of airborne toxicants and particulate matter on our brains. Thus, a third area of interest, for which I am actively performing editorial work for the journal Frontiers in Behavioral Neuroscience, was born