Contact
About James Simpkins
https://medicine.hsc.wvu.edu/neuroscience/faculty-labs/james-w-simpkins-phd/
Positions
Professor
- Organization:
- West Virginia University School of Medicine
- Department:
- Department of Neuroscience
- Classification:
- Faculty
Professor
- Organization:
- West Virginia University School of Medicine
- Department:
- Physiology, Pharmacology & Toxicology
- Classification:
- Faculty
Professor
- Organization:
- West Virginia University School of Medicine
- Department:
- Rockefeller Neuroscience Institute (SOM)
- Classification:
- Faculty
Education
- PhD, Michigan State University
Publications
[2021]
- Quintana DD, Anantula Y, Garcia JA, Engler-Chiurazzi EB, Sarkar SN, Corbin DR, Brown CM, Simpkins JW. (2021). Microvascular degeneration occurs before plaque onset and progresses with age in 3xTg AD mice. Neurobiol Aging.105:115-128.
- Quintana D, Ren X, Hu H, Corbin D, Engler-Chiurazzi E, Alvi M, Simpkins J. (2021). IL-1β Antibody Protects Brain from Neuropathology of Hypoperfusion. Cells. 10(4):855.
- Kim SJ, Russell AE, Wang W, Gemoets DE, Sarkar SN, Simpkins JW, Brown CM. (2021). miR-146a Dysregulates Energy Metabolism During Neuroinflammation. J Neuroimmune Pharmacol.
- Ren X, Hu H, Farooqi I, Simpkins JW. (2021). Author Correction: Blood substitution therapy rescues the brain of mice from ischemic damage. Nat Commun. 12(1):2957.
- Russell AE, Cavendish JZ, Rai A, Vannoy M, Dakhlallah AH, Hu H, Ren X, Amer A, Brown CM, Marsh CB, Simpkins JW, Dakhlallah D. (2021). Intermittent Lipopolysaccharide Exposure Significantly Increases Cortical Infarct Size and Impairs Autophagy. ASN Neuro. 13:1759091421991769.
- Gao X, Boryczka J, Zheng P, Kasani S, Yang F, Engler-Chiurazzi EB, Simpkins JW, Wigginton JG, Wu N. (2021). A "hot Spot"-Enhanced paper lateral flow assay for ultrasensitive detection of traumatic brain injury biomarker S-100β in blood plasma. Biosens Bioelectron. 177:112967.
- Saralkar P, Mdzinarishvili A, Arsiwala TA, Lee YK, Sullivan PG, Pinti MV, Hollander JM, Kelley EE, Ren X, Hu H, Simpkins J, Brown C, Hazlehurst LE, Huber JD, Geldenhuys WJ. (2021). The Mitochondrial mitoNEET Ligand NL-1 Is Protective in a Murine Model of Transient Cerebral Ischemic Stroke. Pharm Res. 38(5):803-817.
[2020]
- Sarvari S, Moakedi F, Hone E, Simpkins JW, Ren X. Mechanisms in blood-brain barrier opening and metabolism-challenged cerebrovascular ischemia with emphasis on ischemic stroke. Metab Brain Dis. 2020 Apr 15. doi: 10.1007/s11011-020-00573-8. [Epub ahead of print] Review. PMID: 32297170
- Ren X, Hu H, Farooqi I, Simpkins JW. (2020). Blood substitution therapy rescues the brain of mice from ischemic damage. Nat Commun. 11(1):4078.
- Dominic D. Quintana, Jorge A. Garcia, Yamini Anantula, Stephanie L. Rellick, Elizabeth B. Engler-Chiurazzi, Saumyendra N. Sarkar, Candice M. Brown and James W. Simpkins, Amyloid-β Causes Mitochondrial Dysfunction via a Ca2+-Driven Upregulation of Oxidative Phosphorylation and Superoxide Production in Cerebrovascular Endothelial Cells. Journal of Alzheimer’s Disease, In Press, 2020.
- Heng Hu,Emily A. Hone, Edward A. P. Provencher, Samuel A. Sprowls, Imran farooqi, Deborah R. Corbin, Saumyendra N. Sarkar, John M. Hollander, Paul R. Lockman, James W. Simpkins & Xuefang Ren, MiR-34a Interacts with Cytochrome c and Shapes Stroke Outcomes, Science reports 10: 3233, 2020.
- Keyana N. Porter, Saumyendra N. Sarkar, Duaa A. Dakhlallah, Dominic D. Quintana, James W. Simpkins, Medroxyprogesterone Acetate Impairs Amyloid Beta Degradation in a Matrix Metalloproteinase-9 Dependent Manner, Frontiers in Neuroscience, in press, 2020.
[2019]
- Saumyendra Sarkar, Ashley Russell, Elizabeth B. Engler-Chiurazzi, Keyana Porter and James W. Simpkins, MicroRNAs and the Genetic Nexus in Aging, Neuroinflammation and Brain Trauma, Aging and Disease 2019 Apr 1;10(2):329-352.
- Ren X, Engler-Chiurazzi EB, Russell AE, Sarkar SN, Rellick SL, Lewis S, Corbin D, Clapper J, Simpkins JW, MiR-34a and stroke: Assessment of non-modifiable biological risk factors in cerebral ischemia. Neurochem Int. 2019 Jul;127: 73-79. PMID: 30365981
- DD Quintana, JA Garcia, SN Sarkar, S Jun, JZ Cavendish, EB Engler-Chiurazzi, Ashley Kerr and JW Simpkins, Hypoxia-reoxygenation of primary astrocytes results in a redistribution of mitochondrial size and mitophagy, Mitochondria 2019 Jul;47:244-255.
- Ashley E. Russell, Sujung Jun, Saumendrya Sarkar, Werner J. Geldenhuys, Sara E. Lewis, Stephanie L. Rellick, and James W. Simpkins, Extracellular vesicles secreted in response to cytokine exposure increase mitochondrial oxygen consumption in recipient cells, Frontiers in Cellular Neuroscience, 2019 Feb 14;13:51. doi: 10.3389/fncel.2019.00051.
- J.Z. Cavendish, S.N. Sarkar, M.A. Colantonio, D.D. Quintana1, N. Ahmed, B.A. White, E.B. Engler-Chiurazzi and J.W. Simpkins, Mitochondrial Movement and Number Deficit in Embryonic Cortical Neurons from 3xTg-AD Mice, J Alzheimers Dis. 2019;70(1):139-151.
- Saumyendra N Sarkar; Elizabeth B Engler-Chiurazzi; John Z Cavendish; Jessica M Povroznik; Ashley E Russell; Dominic D Quintana; Peter H Mathers; James W. Simpkins, Over-expression of miR-34a Induces Rapid Cognitive Impairment and Alzheimer's Disease-like Pathology, Brain Research,. 2019 Jul 8:146327. doi: 10.1016/j.brainres.2019.146327. [Epub ahead of print]
- D. D. Quintana, S. E. Lewis, Y. Anantula, J. A. Garcia, S. N. Sarkar, J. Z. Cavendish, C. M. Brown and J. W. Simpkins, The Cerebral Angiome: High Resolution MicroCT Imaging of the Whole Brain Cerebrovasculature in Female and Male Mice, Neuroimage 2019 Aug 22;202:116109. doi: 10.1016/j. [Epub ahead of print] PMID: 31446129
[2018]
- Gina Sizemore, Brandon Lucke-Wold, Charles Rosen, James W. Simpkins, Sanjay Bhatia, Dandan Sun. Temporal Lobe Epilepsy, Stroke, and Traumatic Brain Injury: Mechanisms of Hyperpolarized, Depolarized, and Flow-through Ion Channels Utilized as Tri-Coordinate Biomarkers of Electrophysiologic Dysfunction. OBM Neurobiology, In Press, 2018. 422.
- Dominic D. Quintana, Xuefang Ren, Heng Hu, Elizabeth B. Engler-Chiurazzi, Stephanie L. Rellic1 , Sara E. Lewis, Jessica Povroznik, James W. Simpkins, and Mohammad Alvi, Gradual Common Carotid Artery Occlusion Results in Cerebral White Matter Pathology in a Novel Model of Cerebrovascular Hypoperfusion, Metabolic Brain Disease, In Press, 2018.
- Charles B. Breckenridge, Chad D. Foradori, Pragati Sawhney Coder, James W. Simpkins, Robert L. Sielken, Jr., Robert J. Handa, Changes in sensitivity to the effects of atrazine on the LH surge in female SD rats after repeated daily doses: Correlation with liver enzyme expression. Birth Defects Research, 110: 246-258, 2018.
- Emily A Hone, Heng Hu, Samuel A. Sprowls, Imran Farooqi, Kimberly Grasmick, Paul R. Lockman, James W. Simpkins, and Xuefang Ren, Biphasic Blood-Brain Barrier Openings after Stroke, Neurological Disorders and Stroke International, In Press, 2018.
- Charles I. Mosimah, Pamela J. Murray, James W. Simpkins, Not All Clots Are Created Equal: A Review of Deficient Thrombolysis with Tissue Plasminogen Activator (tPA) in Patients with Metabolic Syndrome, International Journal of Neuroscience, 129: 612-618, 2018.
- Neil T. Sprenkle, Anirudhya Lahiri, James W. Simpkins & Gordon P. Meares, Endoplasmic reticulum stress is transmissible in vitro between cells of the central nervous system, J Neurochemistry, 148: 516-530, 2018.
- Breckenridge CB, Foradori CD, Sawhney Coder P, Simpkins JW, Sielken RL Jr, Handa RJ, Changes in Sensitivity to the Effects of Atrazine on the Luteinizing Hormone Surge in Female Sprague-Dawley Rats after Repeated Daily Doses: Correlation with Liver Enzyme Expression, Birth Defects Res. 2018 Feb 15;110(3):246-258.PMC5884089
- Kimberly A. Grasmick, Heng Hu, Emily A. Hone, Imran Farooqi, Stephanie L. Rellick, James W. Simpkins, Xuefang Ren. Uncoupling of the Electron Transport Chain Compromises Mitochondrial Oxidative Phosphorylation and Exacerbates Stroke Outcomes. Journal of Neuroinfectious Diseases 9: 1-4, 2018.
[2017]
- Shao-Hua Yang , Wenjun Li , Nathalie Sumien , Michael Forster , James W. Simpkins, Ran Liu. Alternative mitochondrial electron transfer for the treatment of neurodegenerative diseases and cancers: Methylene blue connects the dots, Progress in Neurobiology 157: 273-291, 2017.
- E.B. Engler-Chiurazzi , C.M. Brown, J.M. Povroznik , J.W. Simpkins, Estrogens as neuroprotectants: Estrogenic actions in the context of cognitive aging and brain injury, Progress in Neurobiology 157: 188-211, 2017.
- Elizabeth B. Engler-Chiurazzi, Douglas F. Covey, and James W. Simpkins, A Novel Mechanism of Non-feminizing Estrogens in Neuroprotection, Experimental Gerontology, 94:99-102.
- Shinichi Asano, Grant O'Connell, Kent C. Lemaster, Evan R. DeVallance, Kayla W. Branyan, James W. Simpkins, Jefferson C. Frisbee, Taura L. Barr, Paul D. Chantler, Leukocytes Mediate Stroke Serum-Induced Vascular Dysfunction, Exp Physiol. 2017 Oct 1;102(10):1321-1331. [Epub ahead of print] PMID: 28737253
- Gao X, Zheng P, Kasani S, Wu S, Yang F, Lewis S, Nayeem S, Engler-Chiurazzi EB, Wigginton JG, Simpkins JW, Wu N. Paper-Based Surface-Enhanced Raman Scattering Lateral Flow Strip for Detection of Neuron-Specific Enolase in Blood Plasma. Anal Chem. 2017 Sep 19;89(18):10104-10110. [Epub ahead of print] PMID: 28817769
- Charles B. Breckenridge, Chad D. Foradori, Pragati Sawhney Coder, James W. Simpkins, Robert L. Sielken, Jr., Robert J. Handa, Changes in sensitivity to the effects of atrazine on the LH surge in female SD rats after repeated daily doses: Correlation with liver enzyme expression. Birth Defects Research, In Press, 2017.
- Jinzi Wu; Rongron Li; Wenjun Li; Ming Ren; Nopporn Thangthaeng; Nathalie Sumien; Ran Liu; Shaohua Yang; James W Simpkins; Michael J Forster; Liang-Jun Yan, Administration of 5-methoxyindole-2-carboxylic acid that potentially targets mitochondrial dihydrolipoamide dehydrogenase confers cerebral preconditioning against ischemic stroke injury, Free Radical Biology and Medicine, In press, October, 2017.
[2016]
- Russell, AE, DN Doll, SN Sarkar and JW Simpkins, TNF-α and Beyond: Rapid Mitochondrial dysfunction mediates TNF-α-induced neurotoxicity, Journal of Clinical and Cellular Immunology, 2016, 7:6 DOI: 10.4172/2155-9899.1000467
- S. Sarkar, S. Jun, S. Rellick, D.D. Quintana, J.Z. Cavendish, and J. W. Simpkins, Expression of MicroRNA-34a in Alzheimer’s Disease Brain Targets Genes Linked to Synaptic Plasticity, Energy Metabolism, and Resting State Network Activity, Brain Res. 1646: 139-151, 2016 PMID: 27235866
- Randy Strong, Richard A. Miller, Adam Antebi, Clinton M. Astle, Molly Bogue, Martin Denzel, Elizabeth Fernandez, Kevin Flurkey, Karyn L. Hamilton, Dudley W. Lamming, Martin A. Javors, João Pedro de Magalhães, Joe M. McCord, Benjamin F. Miller, Michael Müller, James F. Nelson, Juliet Ndukum, G. Ed. Rainger, Arlan Richardson, David M. Sabatini, Adam Salmon, James W. Simpkins, Nancy L. Nadon, David E. Harrison, Longer lifespan in male mice treated with a non-feminizing steroid, an antioxidant, an α-glucosidase inhibitor or a Nrf2-inducer, Aging Cell. 2016 Jun 16. doi: 10.1111/acel.12496. [Epub ahead of print] PMID: 27312235
- Sarkar, S, S Jun, S, Rellick, S, and Simpkins, JW, Increased expression of miR-34a in late-onset Alzheimer’s disease (LOAD) brain mechanistically links synaptic plasticity and energy metabolism dysfunction, Brain Res. 1646: 139-151, 2016 PMID: 27235866
- Bukeirat M, Sarkar SN, Hu H, Quintana DD, Simpkins JW, Ren X, MiR-34a regulates blood-brain barrier permeability and mitochondrial function by targeting cytochrome c. J Cereb Blood Flow Metab. 2016 Feb; 36(2):387-92.
- Hu, H., Doll, DA, Sun, J, Lewis, SE, Wimsatt, JH, Kessler, MJ, Simpkins, JW and Ren, X., Post-stroke hypothermia predicts stroke severity and is correlated with mitochondrial impairment in cerebrovascular endothelial cells, Aging and Disease 2016 Jan 2;7(1):14-27
- Blake S Moses, William L Slone, Rebecca Evans, Debbie Piktel, Peggi M Angel , Callee Walsh, Pamela Cantrell , Stephanie Rellick, Karen Martin, James W. Simpkins, Laura F Gibson , Bone marrow microenvironment modulation of acute lymphoblastic leukemia phenotype, Exp Hematol. 2016 Jan;44(1):50-59.e2. doi: 10.1016/j.exphem.2015.09.003.
- Ashley B. Petrone, Grant C. O’Connell, Michael D. Regier, Paul D. Chantler, James W. Simpkins, and Taura L. Barr, The Role of Arginase 1 in Post-Stroke Immunosuppression and Ischemic Stroke Severity, Transl Stroke Res. 2016 Apr;7(2):103-10.
- Engler-Chiurazzi, EB, Brown, CM, Povroznik, J, and Simpkins, JW, Estrogens as Neuroprotectants: Estrogenic Actions in the Context of Cognitive Aging and Brain Injury, Prog Neurobiol. 2016 Feb 15. pii: S0301-0082(15)30063-0. doi: 10.1016/j.pneurobio.2015.12.008. [Epub ahead of print] Review. PMID: 26891883 (IF 9.992).
- Jiahong Sun, Heng Hu, Xuefang Ren, James W. Simpkins, Tert-butylhydroquinone Compromises Survival in Murine Experimental Stroke, Neurotoxicol Teratol. 2016 Jan 28;54:15-21. PMID:26827673
- Engler-Chiurazzi, EB, Stapleton, PA, Stalnaker, JJ, Sarkar, SN, Jun, S, Quintana, DD, Ren, X, Hu, H, Nurkiewicz, TR, McBride, C, Yi, J, Simpkins, JW, The Impact of Prenatal Nanomaterial Exposure on Male Adult Sprague Dawley Rat Behavior and Cognition, J Toxicol Environ Health A. 2016 Apr 19:1-6. [Epub ahead of print] PMID: 27092594
- Jiahong Sun Xuefang Ren, Wen Qi, Dan Yuan & James W. Simpkins, Geissoschizine methyl ether, an alkaloid from Yokukansan, protects oxidative stress-mediated cytotoxicity in neurons through a ‘neuronal Warburg effect, J Ethnopharmacology 2016 Apr 22. pii: S0378-8741(16)30235-5. doi: 10.1016/j.jep.2016.04.034. [Epub ahead of print] PMID: 27114061
- Rellick, S. L., Hu, H., Simpkins, J. W., Ren, X. Evaluation of Bioenergetic Function in Cerebral Vascular Endothelial Cells. J. Vis. Exp.19:(117), e54847, doi:10.3791/54847 (2016). PMID: 27911398
- Ren, X and J. W. Simpkins, Updates of knowledge in stroke, Dana Foundation, Progress in Brain Research, June, 2016.
- Mingxing Qian, Elizabeth B. Engler-Chiurazzi, Sara E. Lewis, Nigam P. Rath, James W. Simpkins, and Douglas F. Covey, Structure–activity studies of non-steroid analogues structurally-related to neuroprotective estrogens, Organic and Bio Molecular Chemistry, 2016 Oct 18;14(41):9790-9805. PMID: 27714297
About James Simpkins
Biographical Sketch
James W. Simpkins received his Ph.D. degree under the mentorship of the pioneering neuroendocrinologist, Joseph Meites at Michigan State University in 1977. He then jointed the University of Florida when he rose through the academic ranks to a full professorship in 1986. At the University of Florida, he was the founding director of the Center for the Neurobiology of Aging, served as Chair of two departments and was appointed the Frank Duckworth Professor of Drug Discovery in 1996. In 2000, he became the Professor and Chair of the Department of Pharmacology & Neuroscience and Founding Director of the Institute for Aging and Alzheimer’s Disease Research at the University of North Texas Health Science Center at Fort Worth. In 2012, he joined the faculty of the Department of Physiology and Pharmacology of West Virginia University and serves as the Founding Director of the Center for Basic and Translational Stroke Research. In 2012, he received the Harman Award for Life-time Achievement for Aging Research from the American Association of Aging. In 2015, he was named the Highland Chair of Stroke/Neurology at the WVU. The author of 420 full length publications, his research has focus on the discovery of novel compounds for the treatment of age-related brain disorders, with a particular focus on Alzheimer’s disease and stroke. His body of research has resulted in 22 issued US and foreign patents. He has mentored numerous PhD students, post-doctoral fellows and junior faculty to research and career success at the University of Florida, the University of North Texas Health Science Center and continued to do so at the West Virginia University.
Additional Info
Our lab studies the causes and potential treatment for Alzheimer’s disease (AD) and stroke in animal models and in human subjects. We are trying to understand the cause of brain damage from AD stroke and to develop treatments based on our understanding of causes. We use rats to induce stroke damage and a variety of techniques, including molecular biology, behavioral assessment and drug interventions to determine the causes and potential treatment for stroke damage. Our work also involves the use of transgenic mouse models, primarily for modeling AD. West Virginia has one of the highest incidences of stroke in the nation and currently therapies to limit stoke damage are limited. Our lab therefore is searching for novel treatments that can limit the damage and/or improve recovery from stroke.
Dana Foundation Blog: Dr. Simpkins on Stroke - http://danablog.org/2014/05/29/james-simpkins-on-stroke/
Research Program
Center for Basic & Translational Stroke Research
Research Interests
- We assess the role of mitochondria in stroke propensity and stroke damage. These studies are aims at identifying the mechanism by which mitochondrial dysfunction contributes to stroke damage and the possibility of targeting drugs to these mitochondrial defects as a therapy for prevention/acute treatment of stroke.
- Assessment of vaious classes of drugs on neuroprotection in stroke and AD. We have a drug discovery program that attempts to discover novel compounds with efficacy in stroke and AD. One such class of compounds we have termed non-feminizing estrogens.
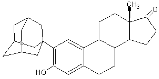

Structure and stroke protective effects of a non-feminizing estrogen (ZYC3)
- We assess the effects of beta amyloid on mitochondrial function to determine how this neurotoxic peptide causes loss of synapses and neurons in the brain.
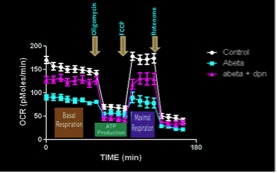
Short-term treatment with beta amyloid (Abeta) causes a profound decline in mitochondrial function in promary neuronal cultures.